 The ripe seed of Physostig'ma veneno'sum Balfour, yielding, by official assay, not less than 0.15 per cent. of alkaloids of Physostigma.
The ripe seed of Physostig'ma veneno'sum Balfour, yielding, by official assay, not less than 0.15 per cent. of alkaloids of Physostigma.
BOTANICAL CHARACTERISTICS.—A lofty, half-shrubby, twining plant, obtaining its name from its peculiar footed stigma. Leaves trifoliate, leaflets ovate. Flowers purplish-pink, in axillary racemes. Legume about 7 inches long.
HABITAT.—Africa.
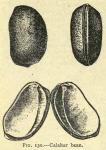
DESCRIPTION OF DRUG.—About the size of a pecan nut, oblong, some what flattened, and kidney-shaped, invested with a light to deep chocolate-brown testa. Along its entire convex edge there extends a prominent black furrow, bordered on each side by a reddish ridge, and traversed the entire length by the raphe as a little ridge in the center. This raphe is terminated at one end by a small funnel-shaped depression, the micropyle. Exalbuminous, embryo large, the cotyledons are concavo-convex, the concave surfaces inclosing a rather large cavity, thus enabling the bean to float upon water. Nearly odorless; taste bean-like, afterward acrid. Spurious calabar beans have been called "calibeans" in European commerce, those occurring the most frequently belonging to the following species: Entada scandens, E. gingalobium D. C., Mucuna urens D. C., and seeds of oil palms, Elaeis Guineensis. E. H. Holmes called attention to certain specimens of calabar beans of commerce bearing a close resemblance to the genuine beans. They were longer, of circular cross-section, and the hilum did not extend the full length of the beans. They also differ chemically, as upon touching the cotyledons with a solution of potassa a permanent yellow tint was produced , and upon treating the spurious article similarly a deep, almost orange, color is formed, turning to a greenish hue. It has been found that the ordinary test-reagents for alkaloids are so sensitive for physostigmine (eserine) that one one-millionth part of a gram may be recognized. The poisonous qualities reside in the seeds, especially in the cotyledons. It has been ascertained that the leaves and stems are not poisonous.
Powder.—Characteristic elements: See Part iv, Chap. I, B.
CONSTITUENTS.—Physostigmine, C15H21N3O2 (also known as eserine), contracting the pupil of the eye; calabarine, a tetanizing principle, a derivative of physostigmine; eseridine, C15H23N3O3 (producing purgation); and physosterin, a neutral principle closely related to cholesterin. These principles are soluble in alcohol. Physostigmine is amorphous, tasteless, reddened by potassa, soda, and lime when exposed to the air, due to absorption of oxygen. The drug sometimes contains over 0-15 per cent. of the alkaloid Physostigmine. Ash, not exceeding 3 per cent.
Preparation of Physostigmine (Eserine).—Treat powdered drug (mixed with 1 per cent. tartaric acid) with water. Shake out coloring matter with ether, make aqueous solution alkaline with an alkaline bicarbonate, and shake out alkaloid with ether. Evaporate ethereal solution.
Preparation of Eseridine (Calabarine).—Precipitate the alkaloid from the liquid from which physostigmine has been separated by lead subacetate and ammonia; evaporate the filtrate, treat the residue with alcohol, precipitate with phosphotungstic acid, and decompose with baryta. It is converted into physostigmine by hydrolysis.
Preparation of Physosterin.—Exhaust beans with petroleum ether and evaporate solvent.
ACTION AND USES.—Physostigmine is used in medicine chiefly for three purposes: as a depressant for the spinal cord; as a stimulant to the intestinal muscles; and to contract the pupils. As a motor depressant physostigmine is useful in the treatment of tetanus and strychnine poisoning. Its greatest value in internal medicine is as a stimulant to intestinal muscles in paralytic forms of colic, but especially in chronic constipation in conjunction with cathartic drugs.
Physostigmine stimulates the secretory nerve-endings of glands and the nerve-endings of striated and smooth muscle. It therefore antagonizes the effects of atropine and curare.
If a drop of 1:200 aqueous solution of eserine is placed in the eye, contraction of the pupil begins in one or two minutes and reaches its maximum in one-half to one hour.
When the alkaloid calabarine is present in excess in the drug, and is taken in overdose, convulsions develop. Dose of drug: 1 to 4 gr. (0.065 to 0.25 Gm.).
OFFICIAL PREPARATIONS.
- Physostigminae Salicylas, Dose: 1/120 to 1/30 gr. (0.0005 to 0.00216 Gm.).
- Extractum Physostigmatis, 1/10 to ½ gr. (0.0064 to 0.0324 Gm.).
- Tinctura Physostigmatis (10 per cent.), 10 to 30 <minim> (0.6 to 2.6 mils).

